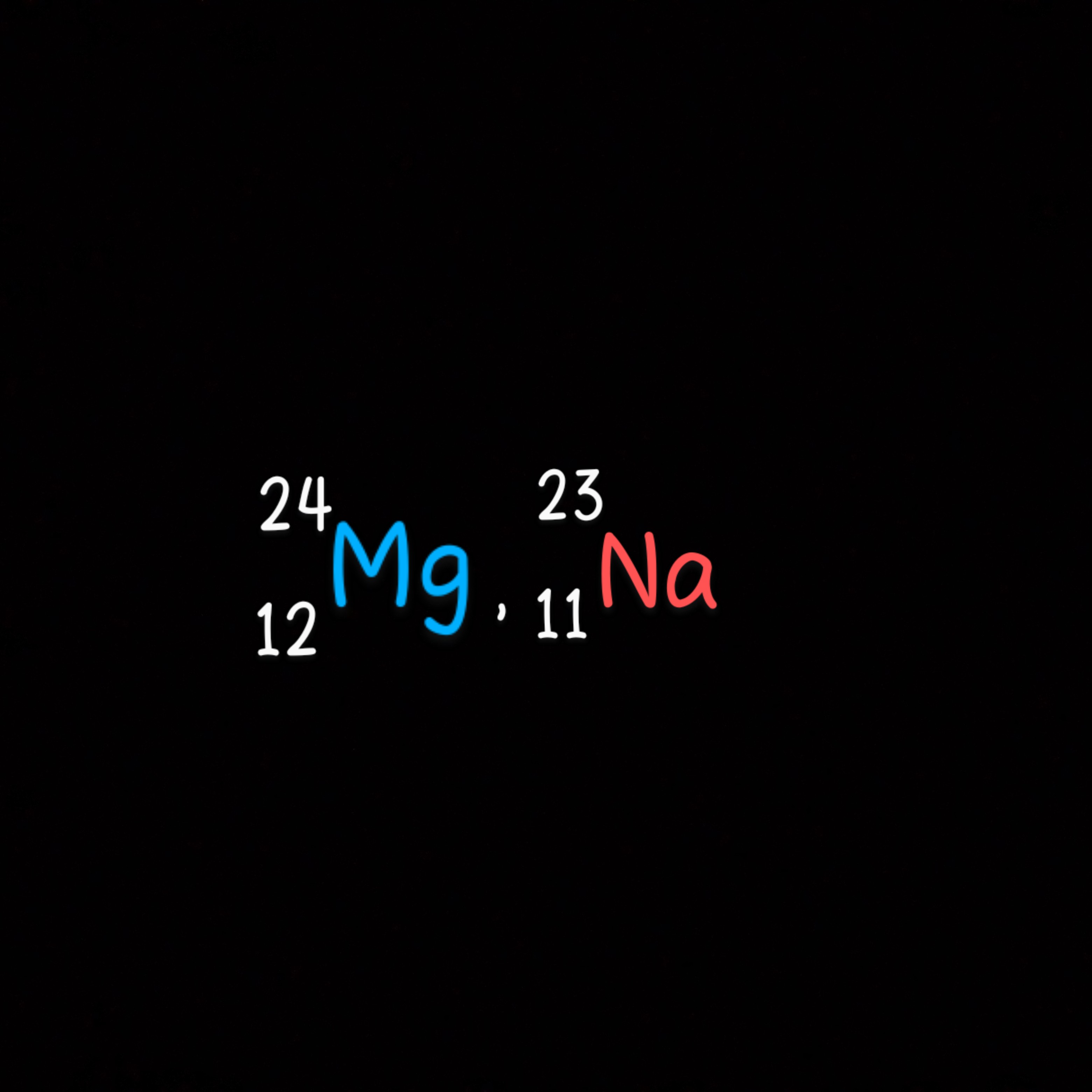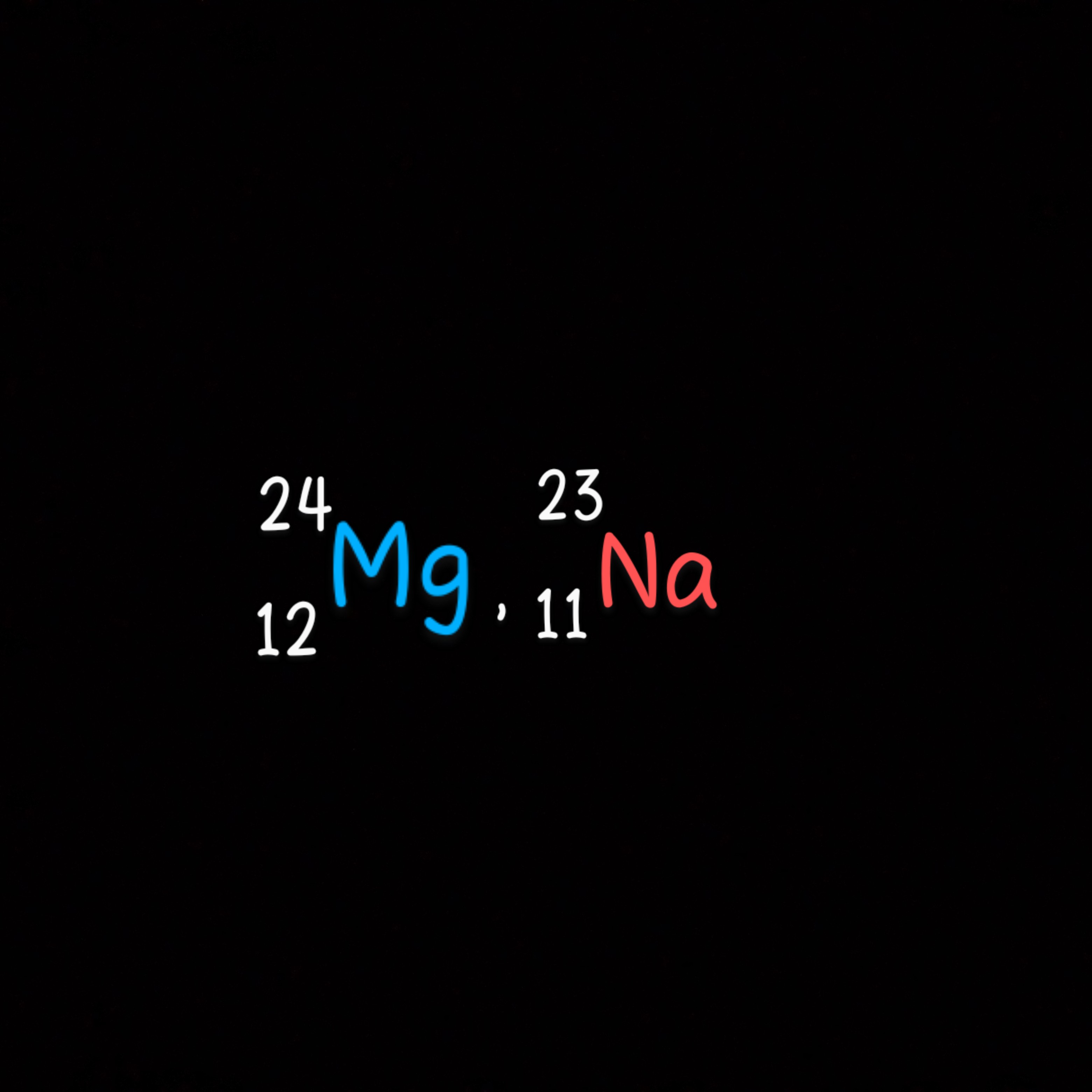
ATOMIC NUMBER AND ATOMIC MASS ( MASS NUMBER). – Atomic number: is the number of protons in the nucleus of an atom . Q. What is the atomic number of elements X with 5 electron in it’s energy levels ? Explain. -Element X has atomic number 5 , this is because it has 5e which equals the number of protons , therefore it has 5p which equals the atomic number. Atomic number= no. of protons= no. of electrons Mass Number ( Atomic mass ) : is the sum of the protons and neutrons in the nucleus of an atom. Mass number= protons (p) + neutrons (n) Q. An element Y has 8 protons , 8 electrons and 8 neutrons, find the mass number of the element Y. Mass number= p+ n = 8+8 = 16. Q. A Magnesium atom has atomic mass of 24 and atomic number 12 , how many neutrons are there in the magnesium atom ? Atomic number = p Atomic mass also known as mass number = p+n Therefore ; neutrons= atomic mass – no. of protons n = 24-12 n= 12. – Both mass number and atomic number can be written along with the symbol of an element. – Mass number is written as the superscript (above the symbol) while atomic number is written as a subscript ( below the symbol) to the left of the chemical symbol .